Uncertainties in Initial Isotope Concentrations
The "clocks-in-the-rocks" approaches make use of the natural radioactive series from long-lived radioisotopes. But rocks formed on the Earth out of a melt would be expected to have some concentration of the daughter products already, and that introduces uncertainty into the determination of the time since the molten state.
It helps that all isotopes are chemically identical, so you know that at the last melting, you had an equilibrium of the isotopes in the melt from which different minerals formed. It also helps that some isotopes are not formed by radioactive decay, so they can act as standards.
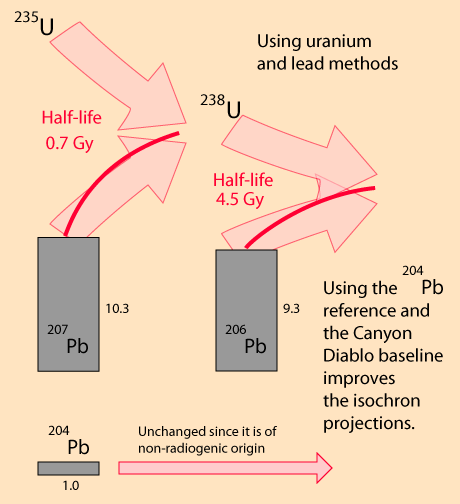 | Over fifty years of intensive study have allowed us to pin down a primordial lead isotope profile, and it is very close to that of meteorites from Canyon Diablo. This standard makes possible the formation of lead-lead isochrons. |
| Outline |
References