The Role of Oxygen in Metabolism
While the provision of energy for metabolism is primarily an oxidation process, that process takes many small steps in cellular respiration. The final step in the chain is taken when the electrons of the chain are delivered to oxygen molecules in the mitochondrial matrix. With the use of four protons and four electrons, the oxygen molecule is converted to two molecules of water. Many steps in the use of food have an oxygen requirement and release carbon dioxide. So the chain of energy events which sustain life start the energy cycle with H2O and CO2, and at the end of the chain produce H2O and CO2.
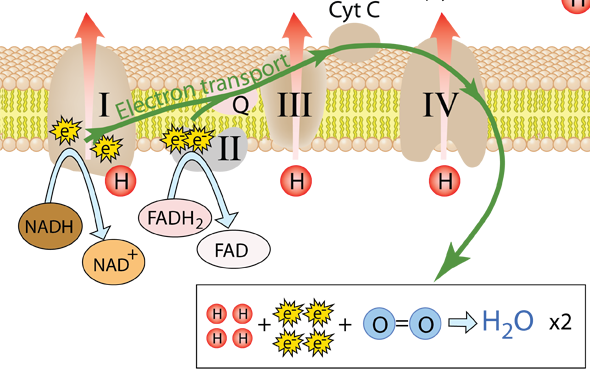
While the steps of the electron transfer chain end with the delivery of the electrons to oxygen to make water, these final steps provide a high population of energetic protons to drive the process of oxidative phosphorylation. That process produces the energetic ATP which enables the continuation of the cycle of life. The close coupling of these two processes demonstrates the crucial role of oxygen in the life process. If there were no oxygen to accept the electrons, then the transport of electrons would come to a halt. Without the electron transport, there would be no proton pumping to create the excess of protons in the inter-membrane space, and with no protons to drive the atp-synthase, no energy would be produced in the form of ATP.
| Energy cycle in living things |
Photosynthesis Concepts
Reference
Moore, et al.
Ch 7
| HyperPhysics***** Biology | R Nave |