Q-cytochrome c oxidoreductase
(Complex III)
The third protein complex in the electron transport chain is named Q-cytochrome c oxidoreductase and is commonly labeled Complex III. It catalyzes the transfer of electrons from the reduced coenzyme Q (ubiquinol) to cytochrome c for transport to Complex IV, the final step in the electron transport chain. For each electron transferred to cytochrome c, two protons are transferred to the intermembrane space to contribute to the process of oxidative phosphorylation to produce the high energy molecule ATP. An interesting scenario plays out in Complex III because of the fact that the reduced coenzyme QH2 provides two electrons, but cytochrome c can receive only one. This leads to a recycling pathway, called the Q cycle, to preserve the second electron for transport.
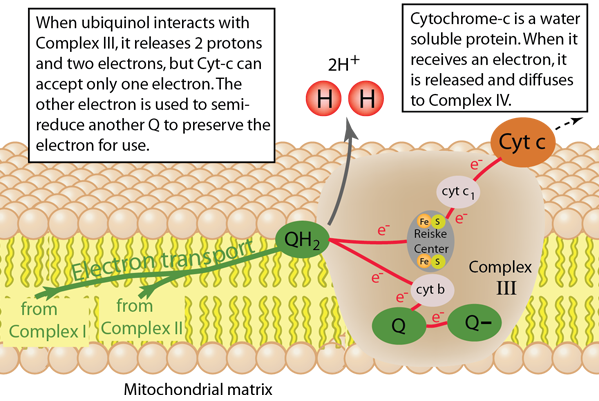
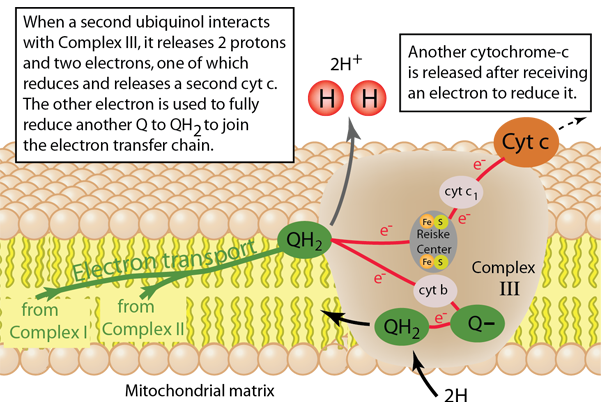
Electrons are collected from Complex I and Complex II and carried by coenzyme Q, which along the way extracts two hydrogens from the matrix to reduce it to form QH2, ubiquinol. QH2 carries two available electrons, but faces the difficulty of transporting them to cytochrome c which can only accept one electron. When QH2 interacts with Complex III it transfers two protons to the intermembrane space, but the two available electrons travel in different paths. Inside Complex III are three important structures. There are two heme-containing structures with iron metal atoms, one called cytochrome c1 with one heme group and another called cytochrome b with two heme groups. The third structure is called the Reiske Center and contains a group 2Fe-2S. The two electrons from QH2 separate, one going to the Reiske Center and one to cytochrome b to start a cyclic process called the Q cycle. The electron traveling to the Reiske Center is transferred to cytochrome c1 and then onto the attached cytochrome c, which is then released to migrate to Complex IV. The second electron travels to cytochrome b where it is used to partially reduce a coenzyme Q (ubiquinone) to the form ubisemiquinone. When another QH2 interacts with Complex III in manner described above, another cyctochrome c receives an electron and the unisemiquinone is fully reduced to QH2 and can join the supply chain described.
| to Complex IV |
| More on Ubiquinol |
Photosynthesis Concepts
Reference
Moore, et al.
Ch 7
Ahern
Biochemistry.., Ch14
Karp
Ch 5.3
AK Lectures, Complex 3
| HyperPhysics***** Biology | R Nave |