Cobalt-60
The cobalt-60 isotope undergoes beta decay with a half-life of 5.272 years.
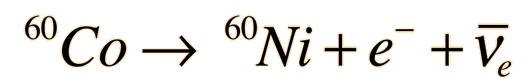
Cobalt-60 decays to Nickel-60 plus an electron and an electron antineutrino. The decay is initially to a nuclear excited state of Nickel-60 from which it emits either one or two gamma ray photons to reach the ground state of the Nickel isotope.
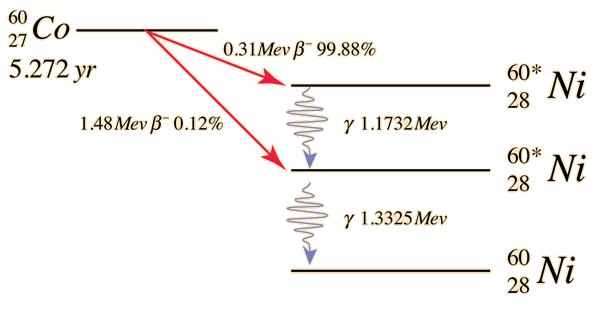
This particular radioisotope is historically important for several reasons. It is involved in the radioactive fallout from nuclear weapons. For many years, the gamma radiation from this decay was the main source for radiation therapy for cancer. This decay was used in the famous experiment by C. S. Wu in which she demonstrated the nonconservation of parity .
| HyperPhysics***** Nuclear | R Nave |