Internal Conversion
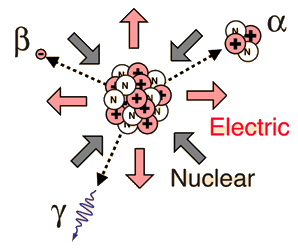
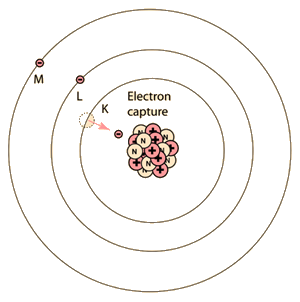
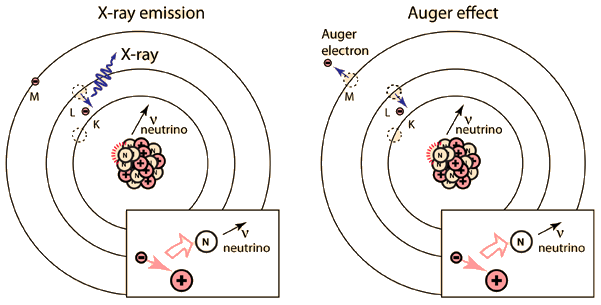
Internal conversion is another electromagnetic process which can occur in the nucleus and which competes with gamma emission. Sometimes the multipole electric fields of the nucleus interact with orbital electrons with enough energy to eject them from the atom. This process is not the same as emitting a gamma ray which knocks an electron out of the atom. It is also not the same as beta decay, since the emitted electron was previously one of the orbital electrons, whereas the electron in beta decay is produced by the decay of a neutron.
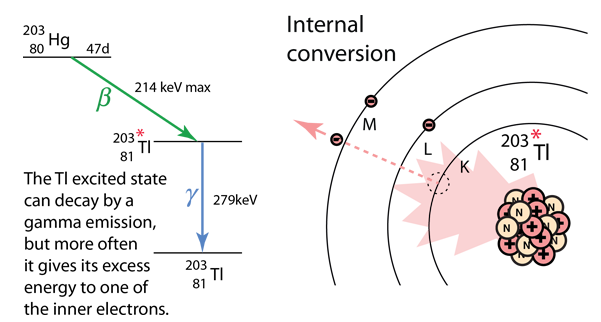
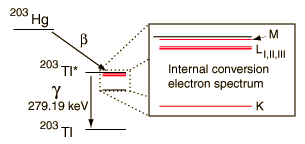
An example used by Krane is that of 203Hg, which decays to 203Tl by beta emission, leaving the 203Tl in an electromagnetically excited state. It can proceed to the ground state by emitting a 279.190 keV gamma ray, or by internal conversion. In this case the internal conversion is more probable. Since the internal conversion process can interact with any of the orbital electrons, the result is a spectrum of internal conversion electrons which will be seen as superimposed upon the electron energy spectrum of the beta emission. The energy yield of this electromagnetic transition can be taken as 279.190 keV, so the ejected electrons will have that energy minus their binding energy in the 203Tl daughter atom.
The diagram above is of course conceptual only and not to scale since the nuclear radius of thallium is modeled to be about 0.7x10-14 m and the radius of the atom is about 1.76x10-10m, a factor of about 25,000 larger! And of course the planetary type orbits of the electrons are unrealistic since the wave properties of the electrons lead to charge distributions that give a finite probability that the K electron shown above will actually extend inside the nucleus so that the nucleus can interact with it and hand off its excess energy. An examination of the electron distribution for the simplest atom, hydrogen, can give the perspective that the electron has a small but finite probability of extending into the nucleus. From the table of binding energies below, you can see that the binding energy of the K-shell electron is over 85,000 electron volts compared to 13.6 eV for the hydrogen electron, or over 6,000 times larger.
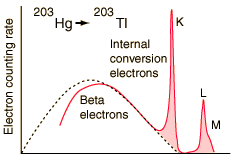 |
Electron emissions from the Hg-203 to Tl-203 decay, measured by A. H. Wapstra, et al., Physica 20, 169 (1954).
|
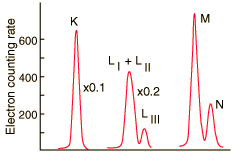 |
At higher resolution, the internal conversion electrons from the L, M and N shells can be resolved. Z. Sujkowski, Ark. Fys. 20, 243 (1961).
|
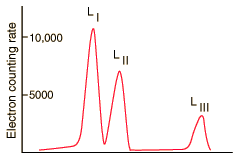 |
At even higher resolution, the three L shells can be resolved. From C. J. Herrlander and R. L. Graham, Nucl. Phys. 58, 544 (1964).
|
The resolution of the electron detection is good enough that such internal conversion electron spectra can be used to study the binding energies of the electrons in heavy atoms. In this case, the measured electron energies can be subtracted from the transition energy as indicated by the gamma emission, 279.190 keV.
Binding energies
for 203Tl |
| K | 85.529 keV |
| LI | 15.347 keV |
| LII | 14.698 keV |
| LIII | 12.657 keV |
| M | 3.704 keV |
|
|
In addition to information from the internal conversion electrons about the binding energies of the electrons in the daughter atom, the relative intensities of these internal conversion electron peaks can give information about the electric multipole character of the nucleus.
|
Index
Reference
Krane
Nuclear Physics, Sec 10.6
Internal conversion Wiki |