Quantum Harmonic Oscillator: Energy Minimum from Uncertainty Principle
The ground state energy for the quantum harmonic oscillator can be shown to be the minimum energy allowed by the uncertainty principle.
The energy of the quantum harmonic oscillator must be at least
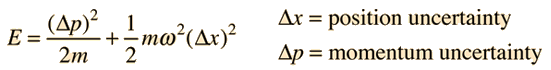
Taking the lower limit from the uncertainty principle
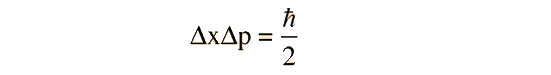
Then the energy expressed in terms of the position uncertainty can be written
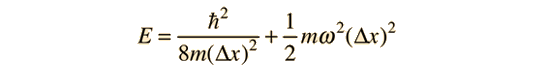
Minimizing this energy by taking the derivative with respect to the position uncertainty and setting it equal to zero gives
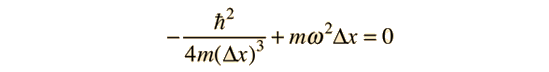
Solving for the position uncertainty gives
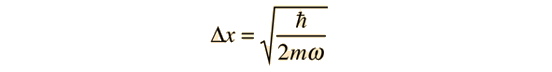
Substituting gives the minimum value of energy allowed.
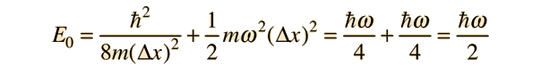
This is a very significant physical result because it tells us that the energy of a system described by a harmonic oscillator potential cannot have zero energy. Physical systems such as atoms in a solid lattice or in polyatomic molecules in a gas cannot have zero energy even at absolute zero temperature. The energy of the ground vibrational state is often referred to as "zero point vibration". The zero point energy is sufficient to prevent liquid helium-4 from freezing at atmospheric pressure, no matter how low the temperature.
Schrodinger equation concepts
Reference
Rohlf
Sec 7-5
| HyperPhysics***** Quantum Physics | R Nave |