Strong Force Coupling Constant
In obtaining a coupling constant for the strong interaction, say in comparison to the electromagnetic force, it must be recognized that they are very different in nature. The electromagnetic force is infinite in range and obeys the inverse square law, while the strong force involves the exchange of massive particles and it therefore has a very short range. It is clear that the strong force is much stronger simply from the fact that the nuclear size (strong force dominant) is about 10-15 m while the atom (electromagnetic force dominant) is about 10-10 m in size. From consideration of the "particle in a box" problem and from just the uncertainty principle, we know it takes greater energy to confine a particle to a smaller volume.
The body of data describing the strong force between nucleons is consistent with a strong force coupling constant of about 1:
But the standard model sees the strong force as arising from the forces between the constituent quarks, which is called the color force. One of the discoveries about this force is that it dimishes inside the nucleons, so that the quarks are able to move freely within the hadrons. The implication for the strong force coupling constant is that it drops off at very small distances. This phenomenon is called "asymptotic freedom" because the quarks approach a state where they can move without resistance in the tiny volume of the hadron. Analysis of the coupling constant with quantum chromodynamics gives an expression for the diminishing coupling constant:
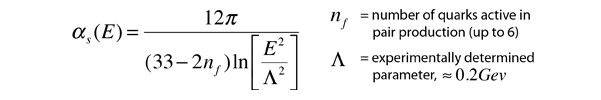
| Coupling constants for fundamental forces |
Fundamental force concepts
Reference
Rohlf
Ch. 1
| HyperPhysics***** Quantum Physics | R Nave |
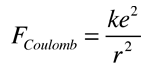
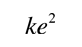
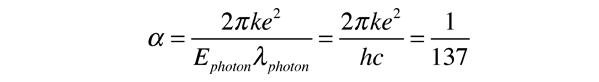
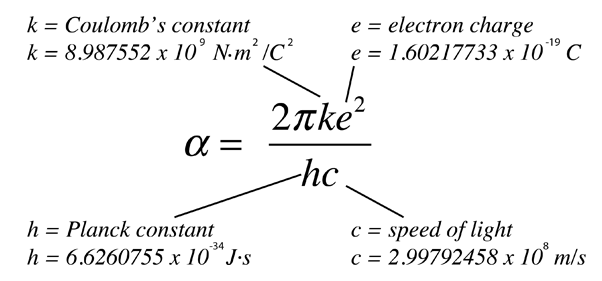
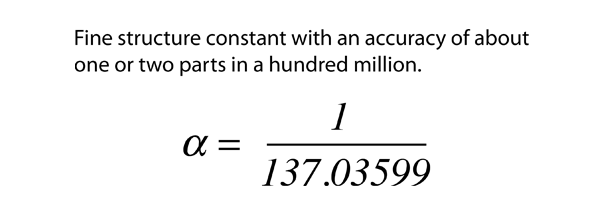
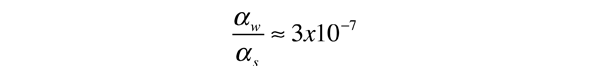
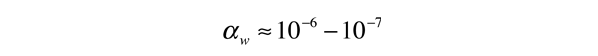
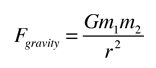
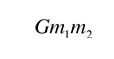
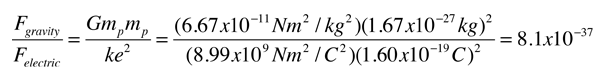 .
.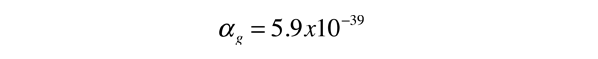
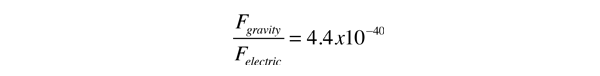 .
.