Helmholtz Free Energy
Four quantities called "thermodynamic potentials" are useful in the chemical thermodynamics of reactions and non-cyclic processes. They are internal energy, the enthalpy, the Helmholtz free energy and the Gibbs free energy. The Helmholtz free energy F is defined by

The internal energy U might be thought of as the energy required to create a system in the absence of changes in temperature or volume. But if the system is created in an environment of temperature T, then some of the energy can be obtained by spontaneous heat transfer from the environment to the system. The amount of this spontaneous energy transfer is TS where S is the final entropy of the system. In that case, you don't have to put in as much energy. Note that if a more disordered (higher entropy) final state is created, less work is required to create the system. The Helmholtz free energy is then a measure of the amount of energy you have to put in to create a system once the spontaneous energy transfer to the sytem from the environment is accounted for.
|
The four thermodynamic potentials are related by offsets of the "energy from the environment" term TS and the "expansion work" term PV. A mnemonic diagram suggested by Schroeder can help you keep track of the relationships between the four thermodynamic potentials. |
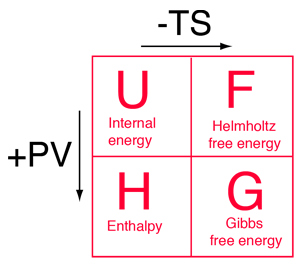 |
Internal energy concepts
Reference
Schroeder
Ch 5
| HyperPhysics***** Thermodynamics | R Nave |
